 This has been necessitated by modest efficacies and shorter persistence rates in initial studies. Within the relative short time of their existence, CAR-Ts have gone through a few generations of evolution. The scFv facilitates the binding of CAR-T cells to their targets (tumor cells), while activation of CAR-T cells is made possible by intracellular domains derived from CD3z ITAM domains. The scFv, derived from a tumor-specific antibody, endows a new antigen specificity to the reconstructed T-cells, allowing for the possible creation of CAR-T cells specific for tumor cells based on the expression of antigens on the surface of tumor cells that can be targeted. CAR is a recombinant receptor construct in which an antibody-derived extracellular single-chain variable fragment (scFv) is linked to intracellular T-cell-signaling domains of the T-cell receptor, enabling the redirection of T-cell-mediated cytotoxicity to cancer cells in an HLA-independent manner. This article focuses on the progress related to CAR-T therapy: the pertinent clinical studies and their major findings, their associated adverse effects, and future perspective.Ĭhimeric Antigen Receptor (CAR)-T therapy has received a lot of interest in recent years. CRS has been addressed by an FDA-approved therapy of its own-tocilizumab. Of particular note are the associated toxicities exemplified by cytokine release syndrome (CRS) and the neurotoxicity. Despite the promising clinical results, there are still several challenges that need to be addressed. This is particularly significant given that the target patient populations of these therapies are those with relapsed and refractory disease who have often progressed on multiple therapies. 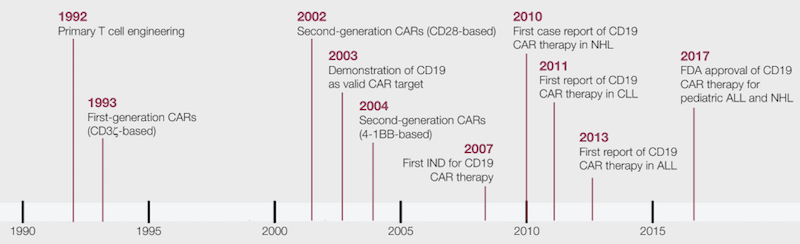 
Over the years, CAR-T cells have evolved to be significantly more persistent in patients’ blood, resulting in a much-improved clinical response and disease remission. Their approval has been a culmination of several phase I and II clinical studies, which are the subject of discussion in this review article. Currently, two CAR-T-cell-based therapies, Tisagenlecleucel and Axicabtagene ciloleucel, are approved by FDA for the treatment of hematological malignancies, acute lymphoblastic leukemia and large B-cell lymphomas. Chimeric Antigen Receptor (CAR)-T cell therapy is an exciting development in the field of cancer immunology, wherein immune T-cells from patients are collected, engineered to create ‘CAR’-T cells, and infused back into the same patient. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
